
Wet adhesion is a challenge for scientists to reproduce in an engineered environment. Certain marine animals like mussels, oysters and barnacles have the natural ability to adhere to surfaces like rock and underwater structures. These animals have the natural adhesion capabilities that enable them to attach to and remain attached to a surface no matter how strong a wave or current is facing them.
Replicating a natural occurring function in nature presents many challenges and the task of creating synthetic wet adhesive materials, has certainly been one of these challenges.
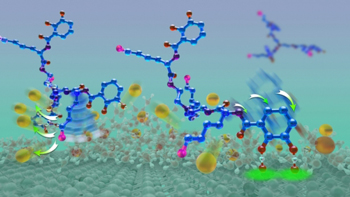
Researchers at the Alison Butler Lab at University of California, Santa Barbara wanted to create a molecular structure that would have adhesive capabilities similar to the chemical composition of mussel foot proteins, so they decided to improve a previously discovered small molecule called the siderophore cyclic trichrysobactin (CTC). What the researchers did to achieve this was modify the molecule and then tested its adhesive strength in aqueous environments. What they created was a compound that very similar with the staying power of mussel glue.
Mussel foot proteins contain similar amounts of lysine and the catechol dopa. Catechols are chemical compounds used in such biological functions as neurotransmission. However, certain proteins have adopted dopa for adhesive purposes.
“There’s real need in a lot of environments, including medicine, to be able to have glues that would work in an aqueous environment,” said co-author Butler, a professor in UCSB’s Dept. of Chemistry and Biochemistry. “So now we have the basis of what we might try to develop from here.”
“We developed a better, more stable molecule than the actual CTC,” Butler explained. “Then we modified it to tease out the importance of the contributions from either lysine or the catechol.”
During the researchers testing, they found only two compounds containing a cationic amine, such as lysine, and catechol exhibited adhesive strength while still having a reduced intervening film thickness. A reduced intervening film thickness is what measures the amount two surfaces can be squeezed together. They found that compounds without catechol had greatly diminished adhesion levels, but similar reduced film thickness. Without lysine, the compounds displayed neither characteristic. “Our tests showed that lysine was key, helping to remove salt ions from the surface to allow the glue to get to the underlying surface,” Maier said.
“In a nutshell, our discovery is that you need lysine and you need the catechol,” Butler concluded. “There’s a one-two punch: the lysine clears and primes the surface and the catechol comes down and hydrogen bonds to the mica surface. This is an unprecedented insight about what needs to happen during wet adhesion.” The researcher’s outstanding results has been published in Science.
—————————————————————————————
Read more in the full article A sticky situation by Julie Cohen of Univ. of California, Santa Barbara here.
Source: Univ. of California, Santa Barbara

Leave a Reply